How to calculate total pressure and partial pressures for gas mixtures from Ideal Gas Law.In a carbon-dating experiment, a particular type of mass spectrometer is used to separate 14 C ^ ) 2.325 × 1 0 − 26 kg ). Total and Partial Pressure - Dalton's Law of Partial Pressures The mole is the SI base unit for an amount of a substance. Mole fraction of water vapor is the ratio of water molecules to air and water molecules. temperature for propane, n-butane, n-heptane and n-pentane hydrocarbons.Ĭhemical, Physical and Thermal Properties of Hydrogen - H 2.Ĭhemical, Physical and Thermal Properties of Methane - CH 4. Molecular WeightĬalculate melting point of hydrocarbons from molecular weight (molar mass). Specific heat at constant volume, specific heat at constant pressure, specific heat ratio and individual gas constant - R - common gases as argon, air, ether, nitrogen and many more. H h/M where: H is the specific enthalpy, kj/kg h is the molar enthalpy, kj/kmol (or J/mol) M is the molar mass of carbon dioxide, 44. Gases - Specific Heats and Individual Gas Constants convert them to the specific property (per unit mass), divide by the molar mass of carbon dioxide (44.010 g/mol). 
Specific gravities of air, ammonia, butadiene, carbon dioxide, carbon monoxide and some other common gases. Molar specific heats for some common gases at constant volume. Gases - Explosion and Flammability Concentration Limitsįlame and explosion limits for gases like propane, methane, butane, acetylene and more. Phase diagram included.Īdiabatic flame temperatures for common fuel gases - propane, butane, acetylene and more - in air or oxygen atmospheres.ĭensities and molecular weights of common gases like acetylene, air, methane, nitrogen, oxygen and others. Isothermal and isentropic gas compression and expansion processes.Ĭhemical, physical and thermal properties of ethylene, also called ethene, acetene and olefiant gas. Temperature and Pressureįigures and table with changes in Prandtl number for carbon dioxide with changes in temperature and pressure.Ĭarbon Dioxide - Thermophysical PropertiesĬhemical, physical and thermal properties of carbon dioxide. We combine (react) many mers of ethylene. Phase diagram included.Ĭhemical, physical and thermal properties of n-Butane.Ĭarbon dioxide - Prandtl Number vs. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) 28. Phase diagram included.ĭry air is a mixture of gases where the average molecular weight (or molar mass) can be calculated by adding the weight of each component.Ĭhemical, physical and thermal properties of benzene, also called benzol. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.Ĭhemical, physical and thermal properties of acetone, also called 2-propanone, dimethyl ketone and pyroacetic acid. 
See also Physical data for hydrocarbons, Physical data for alcohols and carboxylic acids, Physical data for organic nitrogen compounds and Physical data for organic sulfur compounds 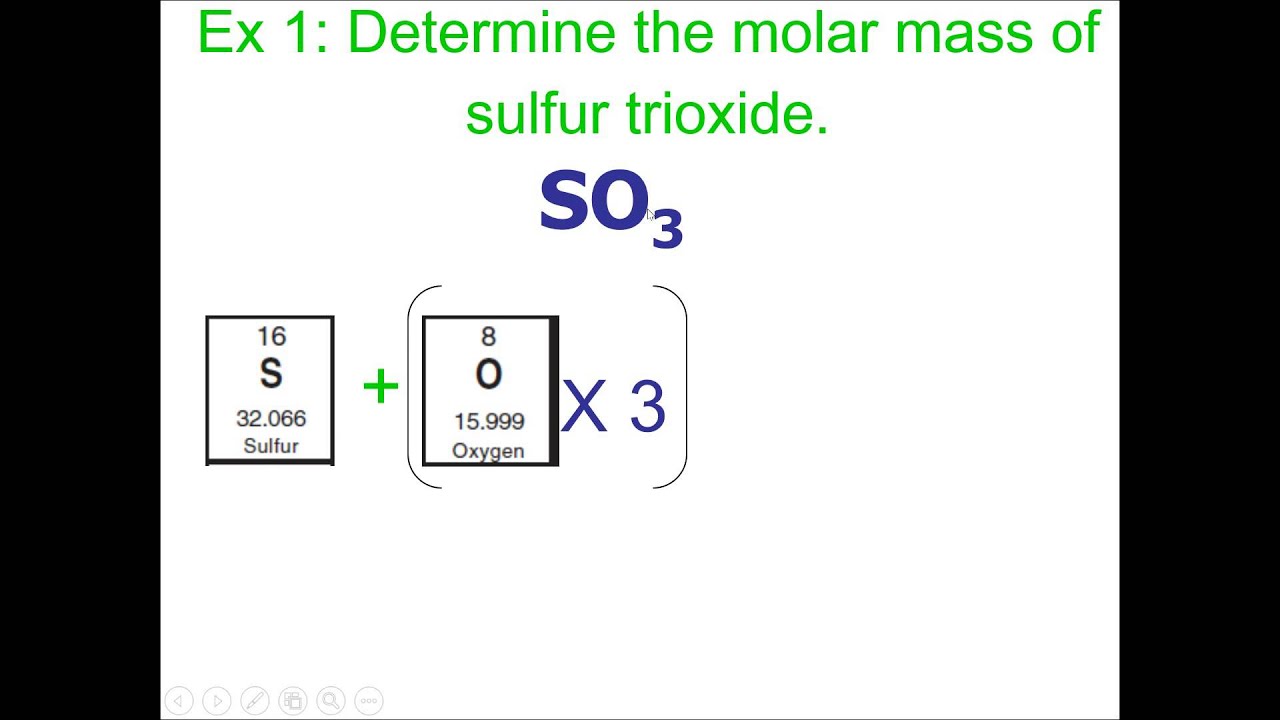
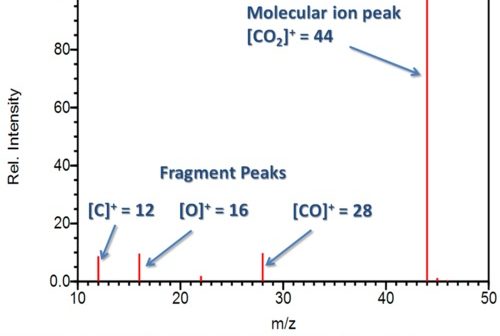
To calculate the molecular weight of ethanol, the molecular weight of each atom in the molecule is summed: The atomic weights of the elements found in organic substances are C = 12.011, H = 1.008, S = 32.065, O = 15.999, and N = 14.007.Įxample: The molecular weight of ethanol (C 2H 5OH) The molecular weight of a pure compound is determined from its chemical formula and the atomic weights of its elements. For this reason, in many cases the unit for the molecular weight is not mentioned however, one must realize that it is not a dimensionless parameter. Molecular weight is represented by the same number in all unit systems regardless of the system used. In the SI system the unit of M is and in the English system the unit is, while in the cgs system the unit of M is. Chemical and physical properties Structure, bonding and molecular vibrations The symmetry of a carbon dioxide molecule is linear and centrosymmetric at its equilibrium geometry. The molecular weight of a substance, also called the molar mass, M, is the mass of 1 mole of that substance, given in M gram. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
